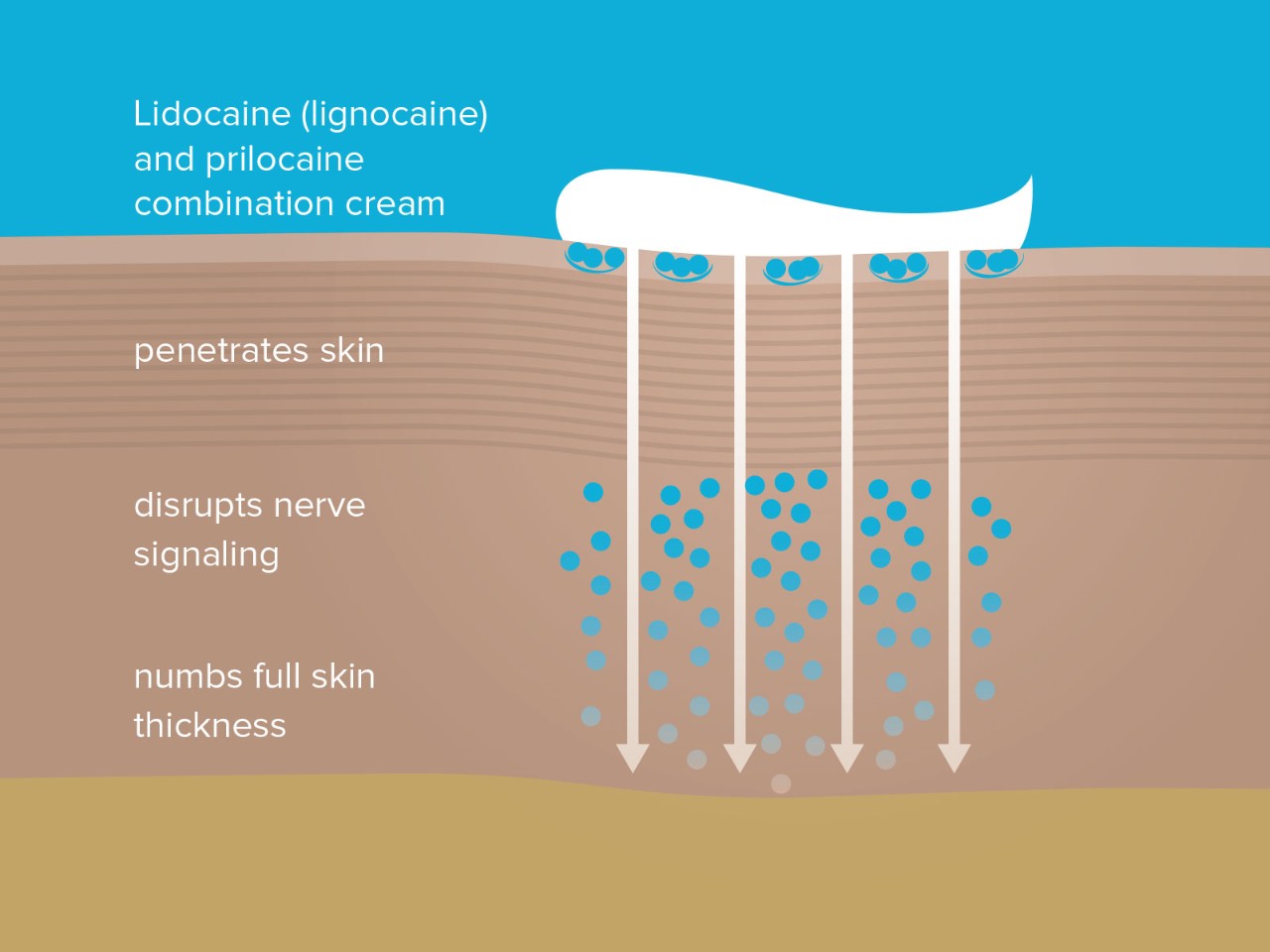
Lidocaine (lignocaine) 2.5% w/w prilocaine 2.5% w/w combination cream can anaesthesia the full skin thickness – over 4 mm deep7,8.
* Price based on discount chemist online purchase prices as per March 30, 2021
1. Britt H, Miller GC, Valenti L, Henderson J, Bayram C, Gordon J. The changing face of Australian general practice across the decades. Aust Fam Physician 2016; 45(9):628–3.
2. Britt H, Miller GC, Henderson J, Bayram C, Harrison C, Valenti L et al. General practice activity in Australia 2014–15. General practice series no. 38. Sydney: Sydney University Press, 2015.
3. Britt H, Miller GC, Charles J, Pan Y, Valenti L, Henderson J. General practice activity in Australia 2005–06. General practice series no. 19. AIHW cat. no. GEP 19. Canberra: Australian Institute of Health and Welfare. 2007.
4. Taddio A, McMurtry CM, Shah V, Riddell RP, Chambers CT, Noel M et al. Reducing pain during vaccine injections: clinical practice guideline. CMAJ 2015; 187(13):975–82.
5. Keogh S, Flynn J, Marsh N, Higgins N, Davies K, Rickard CM. Nursing and midwifery practice for maintenance of vascular access device patency. A cross-sectional survey. Int J Nurs Stud 2015; 52(11):1678–85.
6. ABC. Australia failing to meet best practice guidelines for IV treatment [internet]. 2016 [cited 2018 May 24]. Available from: https://www.abc.net.au/radionational/programs/breakfast/australia-fails-to-meet-best-practice-guidelines-iv-treatment/7379770
7. Bjerring P, Arendt-Nielsen L. Depth and duration of skin analgesia to needle insertion after topical application of Emla cream. Br J Anaesth 1990; 64(2):173–7.
8. NIH. National Cancer Institutes SEER training modules [internet]. 2018 [cited 2018 May 1]. Available from: https://training.seer.cancer.gov/melanoma/anatomy/layers.html
9. US FDA. EMLA® CREAM (lidocaine 2.5% and prilocaine 2.5%).
10. Tadicherla S, Berman B. Percutaneous dermal drug delivery for local pain control. Ther Clin Risk Manag 2006; 2(1):99–113. 11. Numit Product Information. June 2016.
11. Numit Product Information.